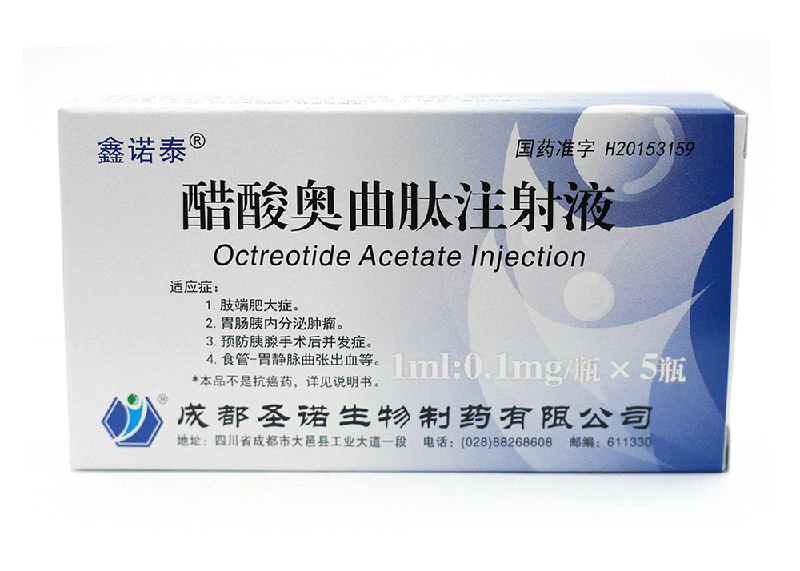
[drug name] general name: polypeptide drug: octreotide acetate injection
Chinese Pinyin: cusuanaoqutai Zhusheye
[ingredients] main ingredients: octreotide acetate is the main ingredient of this product. Chemical name: octreotide acetate, d-phenylalanyl-l-cysteinyl-l-phenylalanyl-d-tryptophan-l-lysine-l-threoniyl-n - [(1R, 2R) - 2-hydroxy-1 - (hydroxymethyl) propyl] - l-cysteinyl ring (2 → 7) - disulfide bond acetate.
Molecular formula: c49h66n10o10s2. Xc2h4o2 molecular weight: 1019.26. × 60.02
Auxiliary materials: lactic acid, mannitol, sodium bicarbonate, water for injection.
[properties] this product is a colorless clear liquid.
[indications] to control the symptoms of patients with acromegaly who cannot fully control the condition of the disease and reduce the plasma levels of growth hormone (GH) and insulin-like growth factor-1 (IGF-1). It can also be used to treat patients with acromegaly who are unable or unwilling to operate, or to treat patients with intermittent acromegaly whose radiotherapy has not yet taken effect.
Related information
[usage and dosage] acromegaly was injected subcutaneously every 8 hours or 12 hours, 0.05-0.1 mg each time, and then adjusted according to the level of circulating GH, IGF-1, clinical response and tolerance (Objective: GH less than 2.5 ng / ml; normal range of IGF).
The optimal daily dose for most patients was 0.2-0.3 mg. GH levels were measured every six months in patients who received the same dose of treatment for a long time. The maximum dose of 1.5 mg per day should not be exceeded. By monitoring the plasma GH level, the dosage can be reduced as appropriate after several months of treatment.
If there is no decrease of GH level and no clinical reaction within one month, the drug should be stopped. Gastrointestinal and pancreatic endocrine tumors were initially subcutaneously injected once or twice a day, 0.05 mg each time. According to the clinical response and the hormone concentration secreted by the tumor (in the case of carcinoid, according to the urine excretion of 5-hydroxyindoleacetic acid) and tolerance, it gradually increased to 0.1-0.2 mg each time, three times a day.
Individual cases may require higher doses. The maintenance dose depends on individual differences. If the clinical symptoms and laboratory tests are not improved within 1 week after the application, the drug should be stopped. To prevent postoperative complications of pancreatic surgery, subcutaneous injection of 0.1 mg three times a day for 7 consecutive days, the first medication at least 1 hour before operation.
0.025mg/h was given intravenously for 5 days at most. Octreotide can be diluted with normal saline. In cirrhotic patients with esophagogastric variceal bleeding, octreotide can be given intravenously for 5 days.
The half-life of drugs in patients with liver dysfunction cirrhosis is prolonged, so the maintenance dose needs to be changed. Renal dysfunction has no effect on the total exposure (AUC) after subcutaneous administration, so it is not necessary to adjust the dosage of octreotide.
[adverse reactions] the most frequently reported adverse reactions during octreotide treatment include gastrointestinal diseases, neurological diseases, hepatobiliary diseases, and metabolic and nutritional diseases. The most common adverse reactions in octreotide clinical trials were diarrhea, abdominal pain, nausea, flatulence, headache, cholelithiasis, hyperglycemia and constipation. Other common adverse reactions were dizziness, local pain, biliary mud formation, thyroid dysfunction (such as decreased TSH, total T4 and free T4), loose stool, impaired glucose tolerance, vomiting, weakness and hypoglycemia. Gastrointestinal diseases in rare cases, gastrointestinal adverse reactions may be similar to acute intestinal obstruction, with progressive abdominal distention, severe upper abdominal pain, abdominal tenderness and muscle tension.
The frequency of known gastrointestinal adverse events decreased with the time of continuous treatment. The occurrence of gastrointestinal adverse reactions can be reduced by avoiding meals before and after administration of octreotide acetate subcutaneously, that is, administration between meals or before sleep. The injection site reflects the pain, tingling or burning sensation of the subcutaneous injection site, with redness and swelling, rarely lasting for more than 15 minutes. Local discomfort can be reduced by bringing the solution to room temperature before injection or by using a concentrated solution to reduce the volume of injection.
Although metabolic and nutritional diseases have detected increased fat excretion in the stool, there is no evidence that long-term use of octreotide can lead to malnutrition due to malabsorption. In very rare cases, acute pancreatitis was reported hours or days before the treatment with octreotide acetate subcutaneous injection, and recovered after discontinuation. In addition, patients who had been treated subcutaneously with octreotide acetate for a long time had reported pancreatitis caused by cholelithiasis. In patients with acromegaly and carcinoid syndrome, ECG changes such as QT interval prolongation, electrical axis shift, early repolarization, low voltage, R / s conversion, early increase of R wave and nonspecific ST-T wave were observed. The relationship between these events and octreotide acetate has not been determined as most of these patients have underlying heart disease (see note).
Adverse reactions in clinical studies are listed in Table 1 of the list of adverse reactions in clinical studies of octreotide. According to the frequency of adverse drug reactions, the following regulations were adopted: very common (≥ 1 / 10), common (≥ 1 / 100, < 1 / 10), rare (≥ 1 / 1000, < 1 / 100), rare (≥ 1 / 10000, < 1 / 1000), very rare (< 1 / 10000), including individual reports. In each frequency group, adverse reactions were ranked in descending order of severity.
Chengdu Shengnuo Biotechnology Co.
Tel: 86-28-88203630
Fax: 86-28-88203630
Email: roleagh@gmail.com
QQ:2539328606
Facebook:[LEI LI](https://www.facebook.com/profile.php? id=100047326162701)
Linkedin: Roleagh
Chengdu Shengnuo Biotechnology Co., Ltd. has "Chengdu polypeptide drug engineering technology research center" in Chengdu, mainly engaged in polypeptide, polypeptide drug and beauty peptide research. Our zero defect has passed the FDA certification, and now it has become the first-class professional peptide drug and product development, technology transfer, technical service and peptide drug industry in the scale production and export of China's parks.